dye laser
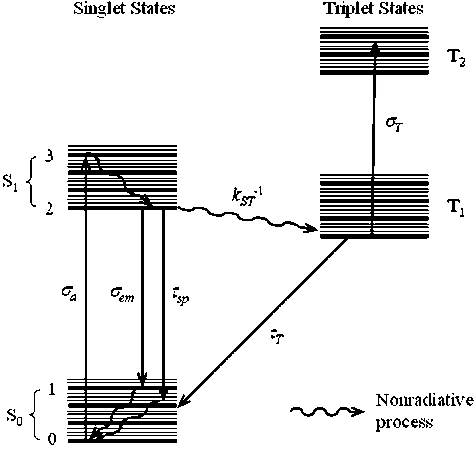
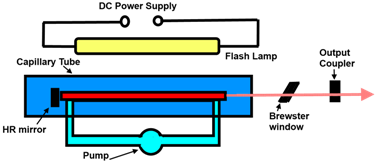
A dye laser is a laser that uses an organic dye as the lasing medium, usually as a liquid solution. Compared to gases and most solid state lasing media, a dye can usually be used for a much wider range of wavelengths, often spanning 50 to 100 nanometers or more. The wide bandwidth makes them particularly suitable for tunable lasers and pulsed lasers. The dye rhodamine 6G, for example, can be tuned from 635 nm (orangish-red) to 560 nm (greenish-yellow), and produce pulses as short as 16 femtoseconds.Moreover, the dye can be replaced by another type in order to generate an even broader range of wavelengths with the same laser, from the near-infrared to the near-ultraviolet, although this usually requires replacing other optical components in the laser as well, such as dielectric mirrors or pump lasers.<br />Close-up of a table-top CW dye laser based on rhodamine 6G, emitting at 580 nm (yellow). The emitted laser beam is visible as faint yellow lines between the yellow window (center) and the yellow optics (upper-right), where it reflects down across the image to an unseen mirror, and back into the dye jet from the lower left corner. My The orange dye-solution enters the laser from the left and exits to the right, still glowing from triplet phosphorescence, and is pumped by a 514 nm (blue-green) beam from an argon laser. The pump laser can be seen entering the dye jet, beneath the yellow window.<br />Dye lasers are used in many applications including:<br />astronomy (as laser guide stars),<br />atomic vapor laser isotope separation<br />manufacturing<br />medicine<br />spectroscopy <br /><br />Some of the laser dyes are rhodamine (orange, 540–680 nm), fluorescein (green, 530–560 nm), coumarin (blue 490–620 nm), stilbene (violet 410–480 nm), umbelliferone (blue, 450–470 nm), tetracene, malachite green, and others. While some dyes are actually used in food coloring, most dyes are very toxic, and often carcinogenic.Many dyes, such as rhodamine 6G, (in its chloride form), can be very corrosive to all metals except stainless steel. Although dyes have very broad fluorescence spectra, the dye's absorption and emission will tend to center on a certain wavelength and taper off to each side, forming a tunability curve, with the absorption center being of a shorter wavelength than the emission center.<br />The figure below shows Typical energy levels of a dye molecul <br /><br />MSc Hawraa Abdalkareem