coordination compound for M.Sc Essa Farhan Deab
23/02/2022 Share :
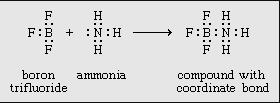
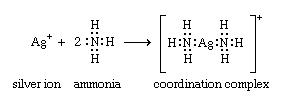
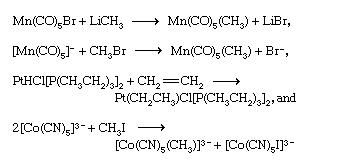
coordination compound, any of a class of substances with chemical structures in which a central metal atom is surrounded by nonmetal atoms or groups of atoms, called ligands, joined to it by chemical bonds. Coordination compounds include such substances as vitamin B12, hemoglobin, and chlorophyll, dyes and pigments, and catalysts used in preparing organic substances. A major application of coordination compounds is their use as catalysts, which serve to alter the rate of chemical reactions. Certain complex metal catalysts, for example, play a key role in the production of polyethylene and polypropylene. In addition, a very stable class of organometallic coordination compounds has provided impetus to the development of organometallic chemistry. Organometallic coordination compounds are sometimes characterized by “sandwich” structures, in which two molecules of an unsaturated cyclic hydrocarbon, which lacks one or more hydrogen atoms, bond on either side of a metal atom. This results in a highly stable aromatic system. with the cobalt(3+) ion and the positive charge is balanced by three chloride anions. The particularly significant feature of this observation was the recognition that two independently stable compounds (i.e., cobalt chloride and ammonia) could combine to form a new chemical compound with properties <br />Structure and bonding of coordination compounds <br />Werner originally postulated that coordination compounds can be formed because the central atoms carry the capacity to form secondary, or coordinate, bonds, in addition to the normal, or valence, bonds. A more complete description of coordinate bonding, in terms of electron pairs, became possible in the 1920s, following the introduction of the concept that all covalent bonds consist of electron pairs shared between atoms, an idea advanced chiefly by the American physical chemist Gilbert N. Lewis. In Lewis’s formulation, when both electrons are contributed by one of the atoms, as in the boron-nitrogen bond formed when the substance boron trifluoride (BF3) combines with ammonia, the bond is called a coordinate bond:<br />In Lewis’s formulas, the valence (or bonding) electrons are indicated by dots, with each pair of dots between two atomic symbols representing a bond between the corresponding atoms.Following Lewis’s ideas, the suggestion was made that the bonds between metals and ligands were of this same type, with the ligands acting as electron donors and the metal ions as electron acceptors. This suggestion provided the first electronic interpretation of bonding in coordination compounds. The coordination reaction between silver ions and ammonia illustrates the resemblance of coordination compounds to the situation in the boron-nitrogen compound. According to this view, the metal ion can be regarded as a so-called Lewis acid and the ligands as Lewis bases:<br />A coordinate bond may also be denoted by an arrow pointing from the donor to the acceptor.<br />Isomerism Coordination compounds often exist as isomers—i.e., as compounds with the same chemical composition but different structural formulas. Many different kinds of isomerism occur among coordination compounds. The following are some of the more common types. Cis-trans isomerism Cis-trans (geometric) isomers of coordination compounds differ from one another only in the manner in which the ligands are distributed spatially; for example, in the isomeric pair of diamminedichloroplatinum compoundsthe two ammonia molecules and the two chlorine atoms are situated next to one another in one isomer, called the cis (Latin for “on this side”) isomer, and across from one another in the other, the trans (Latin for “on the other side”) isomer. A similar relationship exists between the cis and trans forms of the tetraamminedichlorocobalt(1+) ion <br />Principal types of complexes The tendency for complexes to form between a metal ion and a particular combination of ligands and the properties of the resulting complexes depend on a variety of properties of both the metal ion and the ligands. Among the pertinent properties of the metal ion are its size, charge, and electron configuration. Relevant properties of the ligand include its size and charge, the number and kinds of atoms available for coordination, the sizes of the resulting chelate rings formed (if any), and a variety of other geometric (steric) and electronic factors.<br />Organometallic complexes Organometallic complexes are complexes formed between organic groups and metal atoms. They can be divided into two general classes: (1) complexes containing metal-carbon σ bonds and (2) π-bonded metal complexes of unsaturated hydrocarbons—that is, compounds with multiple bonds between carbon atoms Synthesis of coordination compounds The great variety of coordination compounds is matched by the diversity of methods through which such compounds can be synthesized. Complex halides, for example, may be prepared by direct combination of two halide salts (either in the molten state or in a suitable solvent). Palladium chloride and potassium chloride, for example, react to give the complex potassium tetrachloropalladate(2−), as shown in the following equation:<br />Another widely used route to coordination compounds is through the direct combination of a metal ion and appropriate ligands in solution. Thus, the addition of a sufficiently high concentration of ammonia to an aqueous solution of a nickel(2+) salt leads, through a series of reactions (see above Aqua complexes), to the formation of the hexaamminenickel(2+) ion, which can be precipitated, for example, as the sulfate salt, [Ni(NH3)6]SO4. Complexes of metal ions in high oxidation states are sometimes more readily formed by adding the ligands to a solution of the metal ion in a lower oxidation state in the presence of an oxidizing agent. Thus, addition of ammonia to an aqueous solution of a cobalt(2+) salt in the presence of air or oxygen leads to the formation of cobalt(3+)-ammine complexes such as hexaamminecobalt(3+), [Co(NH3)6]3+, and pentaammineaquacobalt(3+), [Co(NH3)5(H2O)]3+, ions. Complexes of metals in low oxidation states, such as the carbonyls of metals in their zero oxidation states, can sometimes be prepared by direct combination of the metal with the ligand, as, for example, in the reaction of nickel metal with carbon monoxide. <br />More commonly, a salt of the metal is reduced in the presence of the ligand. An example of this type of synthesis is the reduction of cobalt carbonate with hydrogen in the presence of carbon monoxide to give bis(tetracarbonylcobalt)<br />Similar procedures are applicable to the synthesis of metal sandwich compounds containing cyclopentadienyl and benzene ligands. Dibenzenechromium, for example, can be prepared from chromic chloride, benzene, and aluminum, as shown in the following equation.<br />Hydrido complexes of transition metals can be prepared by reactions of suitable precursors either with molecular hydrogen or with suitable reducing agents such as hydrazine or sodium borohydride; for example,Transition metal complexes containing metal-carbon bonds can be prepared by a variety of routes, some of the more important of which are illustrated by the following examples (for further treatment of carbonyl synthesis.