Scientific Article by Assistant Huda Rafid Al-Khafaji: "EDTA: Chemical Properties and Its Medical, Industrial, and Laboratory Applications"
09/02/2026 Share :
Introduction
Ethylenediaminetetraacetic acid (EDTA) is one of the most important chemical compounds used in scientific, medical, and industrial fields. It is characterized by its high ability to bind metal ions, making it an effective chelating agent in many applications.
Chemical Structure
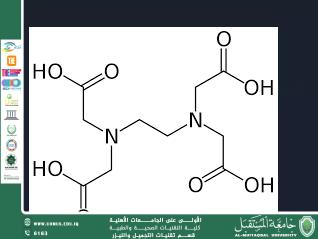
The molecular formula of EDTA is:
C₁₀H₁₆N₂O₈
The molecule consists of:
Two nitrogen atoms.
Four carboxyl groups (-COOH).
This structure enables EDTA to form strong bonds with divalent and trivalent metal ions such as
Calcium (Ca²⁺)
Magnesium (Mg²⁺)
Iron (Fe³⁺)
Copper (Cu²⁺)
Physical and Chemical Properties
EDTA has several important properties, including:
A white crystalline solid.
Poorly soluble in water in its acidic form.
Its solubility increases when converted into salts such as (EDTA-Na₂ or EDTA-Na₄).
Relatively stable under normal conditions.
High ability to form stable complexes with metals.
Mechanism of Action of EDTA
EDTA acts as a chelating agent through the following steps:
Surrounding the metal ion.
Forming multiple coordination bonds.
Creating a stable complex that prevents the metal from reacting with other substances.
This process is known as chelation, and it is the basis of EDTA’s wide range of applications.
Medical Uses
EDTA is used in the medical field in several applications, including:
1. Treatment of Heavy Metal Poisoning
EDTA is used to treat cases of lead and mercury poisoning by binding these metals and facilitating their excretion through urine.
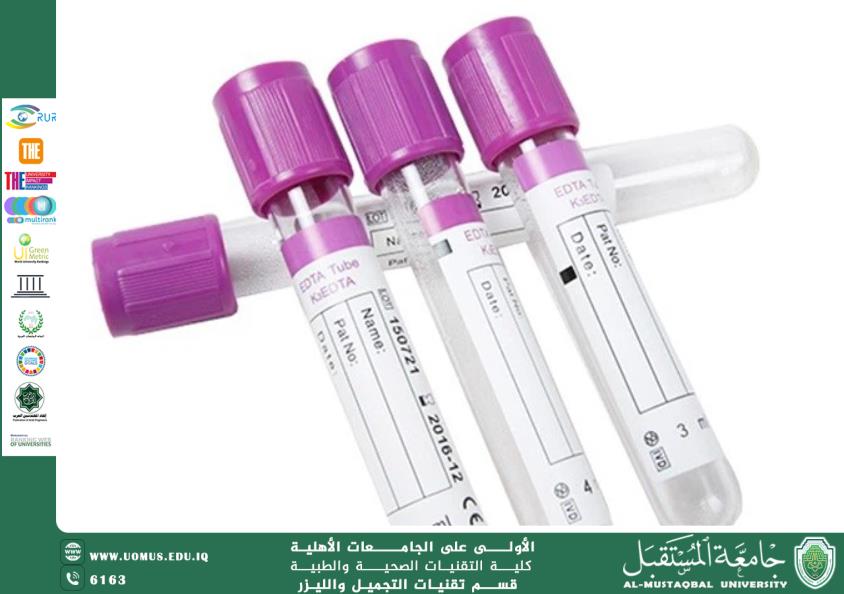
2. Prevention of Blood Coagulation
EDTA is added to blood collection tubes in laboratories to prevent clotting by binding calcium ions, which are essential for the coagulation process.
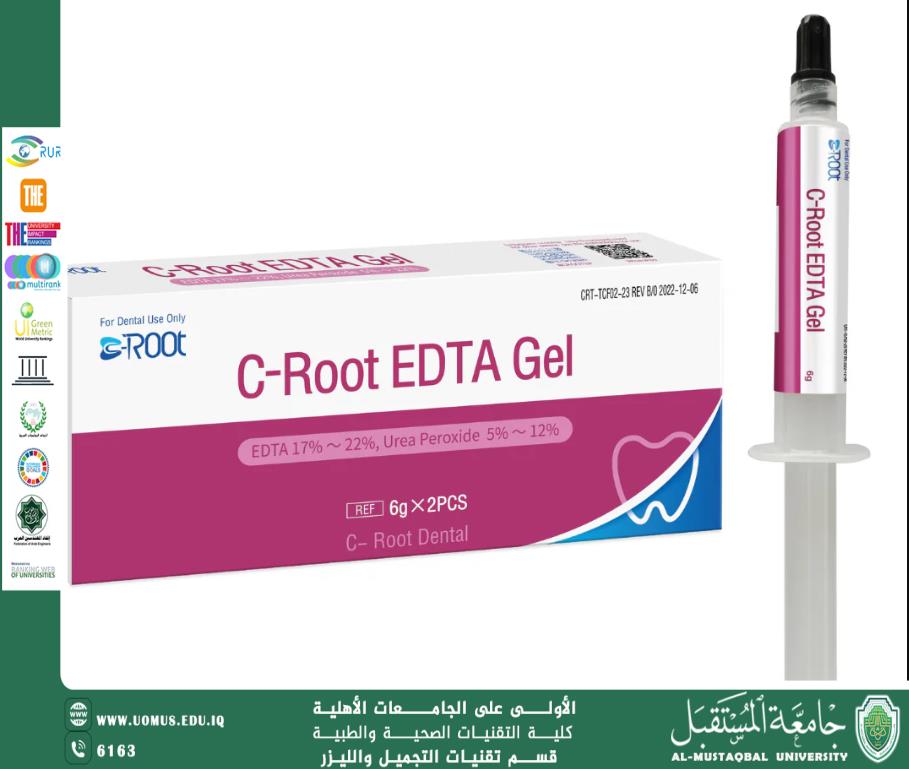
3. Dentistry
It is used in root canal cleaning during endodontic treatment to remove mineral deposits.
Industrial Uses
EDTA is involved in many industrial applications, such as:
Water treatment to remove mineral salts.
Detergent manufacturing to improve effectiveness.
Food industry as a preservative.
Paper and textile industries.
Cosmetics to prevent metal interactions with ingredients.
Laboratory Uses
In scientific laboratories, EDTA is considered an essential substance and is used in:
Complexometric titrations.
Water hardness analysis.
Preservation of biological samples.
Biochemical studies.
Health and Environmental Effects
Health Effects
When used in appropriate doses, EDTA is relatively safe. However, excessive use may lead to:
Deficiency of essential minerals in the body.
Kidney function disorders.
Decreased calcium levels in the blood.
Environmental Effects
EDTA is characterized by its stability in the environment, which may lead to:
Accumulation in water bodies.
Increased mobility of heavy metals in soil.
Potential negative effects on living organisms.
Conclusion
EDTA is an important chemical compound that plays a vital role in medical, industrial, and laboratory fields. Its importance lies in its strong ability to form stable complexes with metals, making it an effective tool in many applications. However, it should be used with caution to avoid potential health and environmental risks.
AL_mustaqbal University is the first university in Iraq