Magnetic Hyperthermia Scientific article By Dr.Mohammed Hashim
19/04/2022 Share :
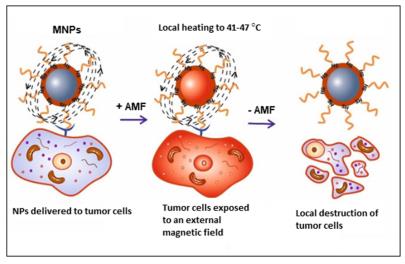
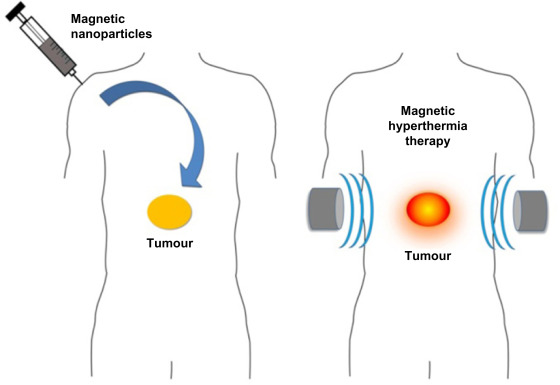
Hyperthermia is the treating of diseases by heating was well-known since the olden era [1]. Increasing the temperature of cells above (41 – 47) C interior of the cell, such as (a) increasing the fluidity and permeability of the cell membrane, (b) slowing down of the mechanisms of synthesis of nucleic acid and protein, (c) inducing protein denaturation and agglomeration, and (d) damaging the tumor vasculature resulting in a reduction of blood flow [2]. Hyperthermia is divided into three main categories depending on the size of the cancer region being treated [3]: whole-body, regional, and local hyperthermia. In the wholebody hyperthermia approach, heats are applied to the whole body in few ways such as using warm water blankets. In the regional hyperthermia method, heat is applied to a whole region of the body using external arrays of applicators. In local hyperthermia, heat is practical to small tumor regions using electromagnetic wave such as microwave, radio wave and ultrasound, which are generated by applicator that is placed at the surface or under skin of superficial cancer. In these methods, the temperature must be increased between (41 and 47) C. All these have its negatives. Some of these negatives [2] are: (a) heat is applied to the healthy cells in addition to the unhealthy ones which could cause negatively side effects, (b) temperature control at the cell level is difficult, (c) the applied heat is not uniform through the targeted region, and (d) the amount of heat delivered is small. In a local hyperthermia, there is a healthier control on the area exposed to heat and a better heat uniformity. However, the local hyperthermia sufferers from two main drawbacks: (a) it is highly invasive for deep cancer regions and (b) the small penetration depth, which is nearly a few centimeters. These drawbacks of local hyperthermia make it healthier used for small and superficial cancer regions. Nanotechnology techniques can help in eliminating these negatives of local hyperthermia. Using local hyperthermia with magnetic nanoparticles (MPs) that are delivered only to the cancer cells and heated externally by an alternating magnetic field (AMF) makes it a noninvasive method, minimizes the side effects and allows for targeting deep cancer cells [4]. The magnetic properties for MPs are determined through two main properties: (a) finitesize effect and (b) surface effect [5]. Finite-size effect is related to the structure of the NPs (single-domain or multi-domain). Instead, surface effects result from several effects such as the symmetry breaking of the crystal structure at the surface of the particle, dangling bonds, oxidation, and surface stain. The role of surface effects increases as the particle size decreases. This is the case because the ratio of the number of surface atoms to the core atoms increases as the particle size decreases. Due to size effect and surface effect, magnetic properties of MPs, such as magnetic moment per atom, saturation magnetization, coercivity, magnetic anisotropy, and Curie temperature, can differ from those of a bulk material [5]. The preferred size of the MPs in most medical applications, is between 10 and 50 nm. Typically, MPs become single domain particle and display superparamagnetic behavior above a certain temperature called the blocking temperature. In the superparamagnetic states, a nanoparticles have a large magnetic moments and performs like a giant paramagnetic atom with the fast response to applied magnetic field with almost zero magnetic reminiscence and coercivity. For magnetic hyperthermia MH application, MPs must possess big saturation magnetization: Ms value that will produce large amounts of heat in the tumor cell under the application of AMF. In addition, large Ms values allow for more control on the movement of the MPs in the blood using external magnetic field [6]. In addition to the requirement of large Ms, MPs should be superparamagnetic to achieve good colloidal stability.<br /><br />References<br />1. Dou, Y.; Hynynen, K.; Allen, C. To heat or not to heat: Challenges with clinical translation of thermosensitive liposomes. J. Control Release 2017, 249, 63–73. <br />2. Mallory, M.; Gogineni, E.; Jones, G.C.; Greer, L.; Simone, C.B. Therapeutic hyperthermia: The old, the new, and the upcoming. Crit. Rev. Oncol. Hematol. 2016, 97, 56–64.<br />3. Fratila, R.; Fuente, J. Nanomaterials for Magnetic and Optical Hyperthermia Applications, 1st ed.; Elsevier: Amsterdam, The Nederland, 2019; pp. 1–10. ISBN 978-0-12-813928-8.<br />4. Dhavalikar, R.; Bohórquez, A.C.; Rinaldi, C. Chapter 10—Image-Guided Thermal Therapy Using Magnetic Particle Imaging and Magnetic Fluid Hyperthermia. In Nanomaterials for Magnetic and Optical Hyperthermia Applications; Micro and Nano Technologies; Fratila, R.M., De La Fuente, J.M., Eds.; Elsevier: Amsterdam, The Netherlands, 2019; pp. 265–286. ISBN 978-0-12-813928-8.<br />5. Obaidat, I.M.; Issa, B.; Haik, Y. Magnetic Properties of Magnetic Nanoparticles for E_cient Hyperthermia.Nanomaterials 2015, 5, 63–89. [CrossRef] [PubMed]<br />6. Carrey, J.; Mehdaoui, B.; Respaud, M. Simple models for dynamic hysteresis loop calculations of magnetic single-domain nanoparticles: Application to magnetic hyperthermia optimization. J. Appl. Phys. 2011, 109, 083921.<br />7. Rajan, A., Sahu, N.K. Review on magnetic nanoparticle-mediated hyperthermia for cancer therapy. J Nanopart Res 22, 319 (2020). https://doi.org/10.1007/s11051-020-05045-9<br />