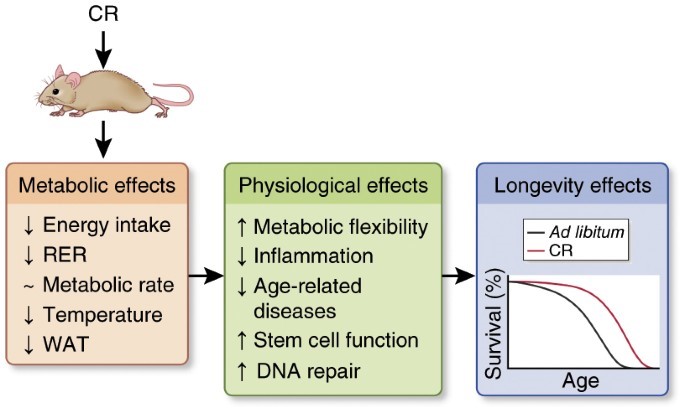
Metabolism and Aging:
27/11/2024 Share :
A link between metabolism and longevity was first proposed by Rubner (Rubner, 1883). From measurements on five different mammals (guinea pigs, cats, dogs, cattle, and horses), he observed that long-lived species are bigger and spend less energy per gram of tissue mass relative to short-lived species. The assumption of such link was rooted from the idea that animals have a limited supply of energy and the quicker they utilise it, the sooner they will die. This idea later became a principle of the rate of living theory (ROL, (Pearl, 1928). In line with this model, previous research on mice, and Glanville fritillary butterflies (Melitaea cinxia) revealed that individuals with higher metabolic rates, corrected for body mass, had a longer life span due to activation of uncoupling proteins(Niitepõld & Hanski, 2013; Wiersma et al., 2004). Similar findings were also reported in mice in which the activation of uncoupling proteins by both chemical and genetic approaches, caused higher metabolic rates and increased longevity (Caldeira da Silva et al , 2008; Keipert et al, 2011). Many other studies on different species have also shown that life span was positively correlated with resting metabolic rate or daily energy metabolism ((Lin et al., 2002; Oklejewicz & Daan, 2002; J R Speakman et al., 2003; Wiersma et al., 2004). However, other studies found no relationship between metabolism and longevity measured in voles ((Selman et al., 2008), blowflies Calliphora stygia ((Hulbert et al, 2004b), Drosophila melanogaster ((Hulbert et al. 2004a). Experiments where metabolism was manipulated by a cold exposure have failed to have impacts on both oxidative stress and longevity in short tailed field voles (Microtus agrestis) (Selman et al., 2008) and on longevity in mice (Vaanholt et al, 2008). Increased metabolism through exercise also had no effects on longevity in mice ((Vaanholt, et al, 2010). However, some other studies where energy expenditure has been manipulated have revealed a negative relationship between energy expenditure and life span (Daan, et al, 1996; Lemon & Barth, 1992), consistent with the predictions of the ROL theory. It has been demonstrated that most of the previous studies examining the link between metabolism and life span across species were performed using inappropriate statistical analysis, in which a correction for body size and phylogenetic independence effects were not applied (Furness & Speakman, 2008; Speakman, 2005).<br /> <br />The metabolic regulation of aging<br /> Witten by: <br />Assistant Professor Dr. Aqeel Handil Al Jothery (PhD UK, Physiology) Anaesthesia Techniques Department, College of Health and Medical Technologies, Al Mustaqbal University, Hilla, Iraq<br /><br />References,<br />Rubner, M. (1883). Uber den Einfluss der Korpergrosse auf Stoff- und Kraft wechsel. Z. Biol., 19, 535–562.<br />Pearl, R. (1928). The rate of living. London, UK: University of London Press.<br />Niitepõld, K., & Hanski, I. (2013). A long life in the fast lane: positive association between peak metabolic rate and lifespan in a butterfly. Journal of Experimental Biology, 216, 1388–1397. http://doi.org/10.1242/jeb.080739<br />Wiersma, P., & Verhulst, S. (2005). Effects of intake rate on energy expenditure, somatic repair and reproduction of zebra finches. The Journal of Experimental Biology, 208(Pt 21), 4091–8. http://doi.org/10.1242/jeb.0185<br />Lin, S.-J., Kaeberlein, M., Andalis, A. A., Sturtz, L. A., Defossez, P.-A., Culotta, V. C., … Guarente, L. (2002). Calorie restriction extends Saccharomyces cerevisiae lifespan by increasing respiration. Nature, 418, 336–340. http://doi.org/10.1038/nature00915 <br />Oklejewicz, M., & Daan, S. (2002). Enhanced Longevity in Tau Mutant Syrian Hamsters, Mesocricetus auratus. Journal of Biological Rhythms, 17, 210–216. http://doi.org/10.1177/07430402017003004<br />Sanz, A., Caro, P., Ibañez, J., Gómez, J., Gredilla, R., & Barja, G. (2005). Dietary restriction at old age lowers mitochondrial oxygen radical production and leak at complex I and oxidative DNA damage in rat brain. Journal of Bioenergetics and Biomembranes, 37, 83–90. http://doi.org/10.1007/s10863-005-4131-0 <br />Selman, C., McLaren, J. S., Mayer, C., Duncan, J. S., Collins, A. R., Duthie, G. G., … Speakman, J. R. (2008). Lifelong alpha-tocopherol supplementation increases the median life span of C57BL/6 mice in the cold but has only minor effects on oxidative damage. Rejuvenation Research, 11, 83–96. http://doi.org/10.1089/rej.2007.0586<br />Hulbert, a J., Clancy, D. J., Mair, W., Braeckman, B. P., Gems, D., & Partridge, L. (2004a). Metabolic rate is not reduced by dietary-restriction or by lowered insulin/IGF-1 signalling and is not correlated with individual lifespan in Drosophila melanogaster. Experimental Gerontology, 39, 1137–1143. http://doi.org/10.1016/j.exger.2004.04.006<br />Hulbert, a J., Usher, M. J., & Wallman, J. F. (2004b). Food consumption and individual lifespan of adults of the blowfly, Calliphora stygia: a test of the “rate of living” theory of aging. Experimental Gerontology, 39, 1485–1490. http://doi.org/10.1016/j.exger.2004.08.008<br />Vaanholt, L. M., Speakman, J. R., Garland, T., Lobley, G. E., & Visser, G. H. (2008). Protein synthesis and antioxidant capacity in aging mice: effects of long-term voluntary exercise. Physiological and Biochemical Zoology, 81, 148–57. http://doi.org/10.1086/525289 <br />Vaanholt, L. M., Daan, S., Garland, T., & Visser, G. H. (2010). Exercising for life? Energy metabolism, body composition, and longevity in mice exercising at different intensities. Physiological and Biochemical Zoology, 83, 239–251. http://doi.org/10.1086/648434<br />Daan, S., Deerenberg, C., & Dijkstra, C. (1996). Increased daily work precipitates natural death in the kestrel. Journal of Animal Ecology, 65, 539–544. <br />Speakman, J. R. (2005). Correlations between physiology and lifespan--two widely ignored problems with comparative studies. Aging Cell, 4, 167–175. http://doi.org/10.1111/j.1474-9726.2005.00162.x<br />