The Nucleus of Radioactive Elements: Structure, Properties, and Applications By: Ahmed Abdul-Salam Muhjam
22/02/2025 Share :
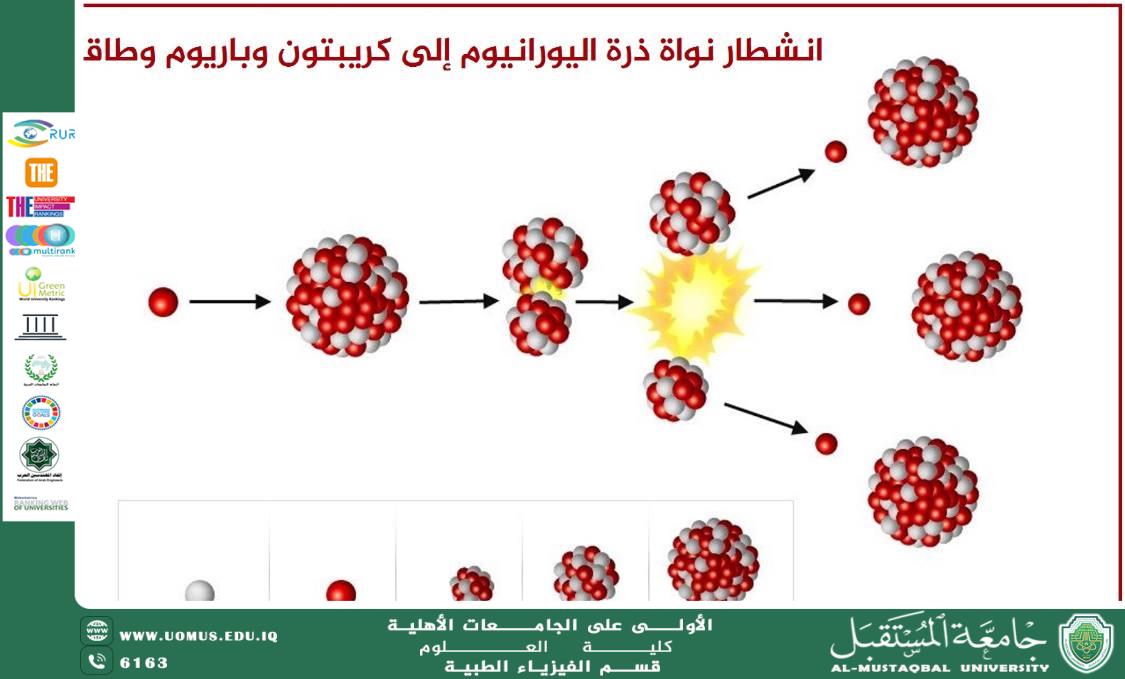
Introduction<br />The atomic nucleus represents the fundamental part of matter, composed of protons and neutrons bound together by the strong nuclear force. In some elements, the nucleus is unstable, leading to the phenomenon of radioactivity, where the nucleus decays spontaneously, emitting radiation in the form of particles and electromagnetic waves. These elements are known as radioactive elements, and they play a pivotal role in various scientific, industrial, and medical applications.<br /><br />Structure of Radioactive Element Nuclei<br />The nucleus of radioactive elements consists of:<br /><br />Protons: Positively charged particles that determine the identity of the element.<br />Neutrons: Electrically neutral particles that contribute to the stability of the nucleus.<br />Strong Nuclear Force: A powerful force that binds protons and neutrons together within the nucleus, overcoming the electromagnetic repulsion between protons.<br />Radioactive elements have an unstable ratio of protons to neutrons, which causes the nucleus to spontaneously decay by emitting alpha or beta particles or gamma rays in an attempt to reach a more stable state.<br /><br />Types of Radioactivity<br />1. Alpha Radiation (α):<br />Consists of alpha particles (helium-4 nuclei: two protons and two neutrons).<br />Weak penetration, easily stopped by a sheet of paper or a thin layer of air.<br />Example: The decay of Uranium-238 into Thorium-234.<br />2. Beta Radiation (β):<br />Consists of electrons or positrons emitted from the nucleus when a neutron transforms into a proton (beta-minus decay) or a proton transforms into a neutron (beta-plus decay).<br />Greater penetration than alpha radiation, blocked by a thin layer of aluminum.<br />Example: The decay of Carbon-14 into Nitrogen-14.<br />3. Gamma Radiation (γ):<br />High-energy electromagnetic radiation emitted from an unstable nucleus.<br />Highly penetrating and can only be blocked by dense materials such as lead or concrete.<br />Example: Gamma-ray emission from Cobalt-60.<br />Radioactive Decay and Half-Life<br />Each radioactive element decays at a specific rate, measured by its half-life—the time required for half of the radioactive material to decay. The half-life varies from one element to another:<br /><br />Carbon-14: Half-life of about 5,730 years, used for dating fossils.<br />Uranium-238: Half-life of about 4.5 billion years, used for dating rocks and planets.<br />Plutonium-239: Half-life of about 24,100 years, used in nuclear reactors and weapons.<br />Scientific and Technological Applications of Radioactive Elements<br />1. Nuclear Medicine:<br />Iodine-131 is used for treating thyroid disorders.<br />Technetium-99m is utilized in medical imaging to detect diseases.<br />2. Nuclear Energy:<br />Uranium-235 and Plutonium-239 are used in nuclear reactors to produce electrical energy.<br />Controlled nuclear reactions provide a long-term energy source.<br />3. Geological and Environmental Detection:<br />Carbon-14 is used for dating fossils and archaeological artifacts.<br />Radioactive isotopes help detect environmental pollution.<br />4. Industrial Applications:<br />Cobalt-60 is used to sterilize medical equipment and preserve food.<br />Radioactive sources are employed in measuring the thickness of metals and materials in heavy industries.<br />Safety and Precautions in Handling Radioactive Elements<br />Due to the hazards of nuclear radiation, appropriate safety measures must be taken, including:<br /><br />Using radiation shields made of lead or concrete to protect against harmful emissions.<br />Minimizing exposure time to radiation whenever possible.<br />Complying with laws and regulations governing the handling of radioactive materials to ensure public safety.<br />Conclusion<br />The nucleus of radioactive elements plays a vital role in nuclear physics and modern technology, contributing significantly to fields such as medicine, energy, industry, and scientific research. However, dealing with these materials requires caution and preventive measures to ensure their safe and sustainable use.<br /><br />University of the Future – The First University in Iraq