Gene Therapy: The Link Between Pharmacology and Molecular Biology By Dr. Qasim Abdul Abbas Zukam, College of Pharmacy, Al-Mustaqbal University
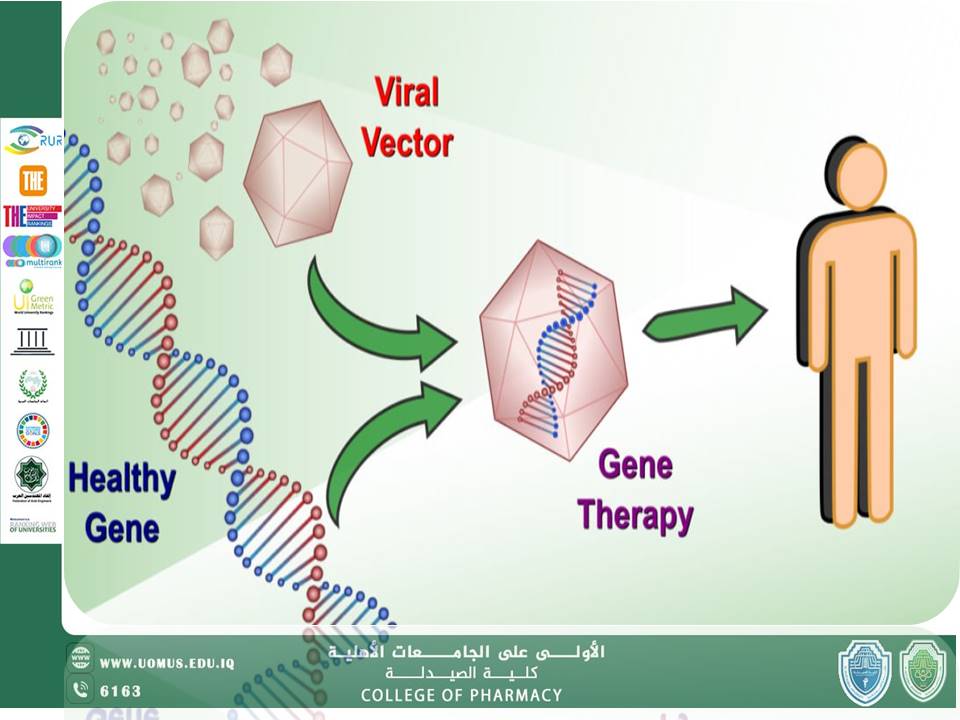
Gene therapy is considered one of the most exciting advancements in modern medicine, offering prospects for treating—or even eradicating—many genetic and acquired diseases. By directly targeting genes, this approach is transforming the future of healthcare and offering hope to millions around the world.<br />Gene therapy involves the modification of genetic material within human cells with the aim of correcting or compensating for faulty genes. This can be achieved by replacing a defective gene, disabling it, or introducing a new gene to help the body resist disease.<br />The concept of gene therapy began to take shape during the 1960s and 1970s, with the first experimental attempts at genetic modification. The first successful human trial was conducted in 1990 to treat Severe Combined Immunodeficiency (SCID). Since then, advances in molecular biology and vector technologies have greatly enhanced the capabilities and opportunities of this field.<br />Gene therapy relies on several core principles:<br />• Vectors: Often modified viruses carrying the therapeutic gene, or non-viral delivery methods such as liposomes or nanoparticles.<br />• Target Cells: Identifying and delivering the gene to specific tissues or cells.<br />• Genetic Modification: Either inserting the gene into the host genome or enabling its effective expression.<br />There are two main types of gene therapy:<br />1. Somatic Gene Therapy: Targets non-reproductive (somatic) cells. The modifications do not pass on to future generations.<br />2. Germline Gene Therapy: Targets reproductive cells (sperm or egg), with modifications that may be inherited by future generations—a topic of both ethical and technical sensitivity.<br />The journey from concept to clinical application of gene therapy involves several critical phases:<br />1. Research and Development<br />Understanding disease-causing genes is fundamental. Researchers identify genes linked to diseases and develop safe and effective vectors for gene delivery.<br />2. Preclinical Studies<br />Laboratory tests on cells and animals evaluate safety, dosage, and efficacy before proceeding to human trials.<br />3. Clinical Trials<br />These are conducted in stages:<br />o Phase I: Focuses on safety and dosage<br />o Phase II: Evaluates effectiveness and side effects<br />o Phase III: Large-scale trials comparing the new therapy with existing treatments<br />Successful trials lead to formal approval.<br />4. Regulatory Approval and Commercialization<br />Regulatory bodies such as the FDA review all data before approving treatments. After approval, the therapy is manufactured, distributed, and subjected to long-term safety monitoring.<br />Current Applications<br />Today, gene therapy is being used to treat many conditions, including:<br />• Inherited Disorders: Such as hemophilia, Duchenne muscular dystrophy, and hereditary blindness.<br />• Cancer: For example, CAR-T cell therapy, in which immune cells are engineered to attack tumors.<br />• Viral Infections: Ongoing trials targeting HIV and other viruses.<br /><br />Gene therapy embodies the spirit of innovation in medicine and pharmacy. For professionals in pharmacy, understanding this field is essential, as it holds immense potential to revolutionize the future of therapeutics.<br /><br />Al-Mustaqbal University – The First Private University in Iraq<br />College of Pharmacy – Leading Among Private Colleges<br />