The Role of Analytical Ultracentrifugation in Studying Protein Aggregation Disorders as Biomarkers of Neurodegenerative Diseases
Neurodegenerative diseases represent some of the most complex disorders at the molecular level. A central pathological hallmark of many of these conditions is protein misfolding, which leads to the formation of abnormal aggregates within or around neuronal cells. Importantly, protein aggregation is not a single-step process; rather, it progresses through intermediate stages that may exert distinct biological effects.
Among the most prominent disorders associated with abnormal protein aggregation are:
Alzheimer's disease
Parkinson's disease
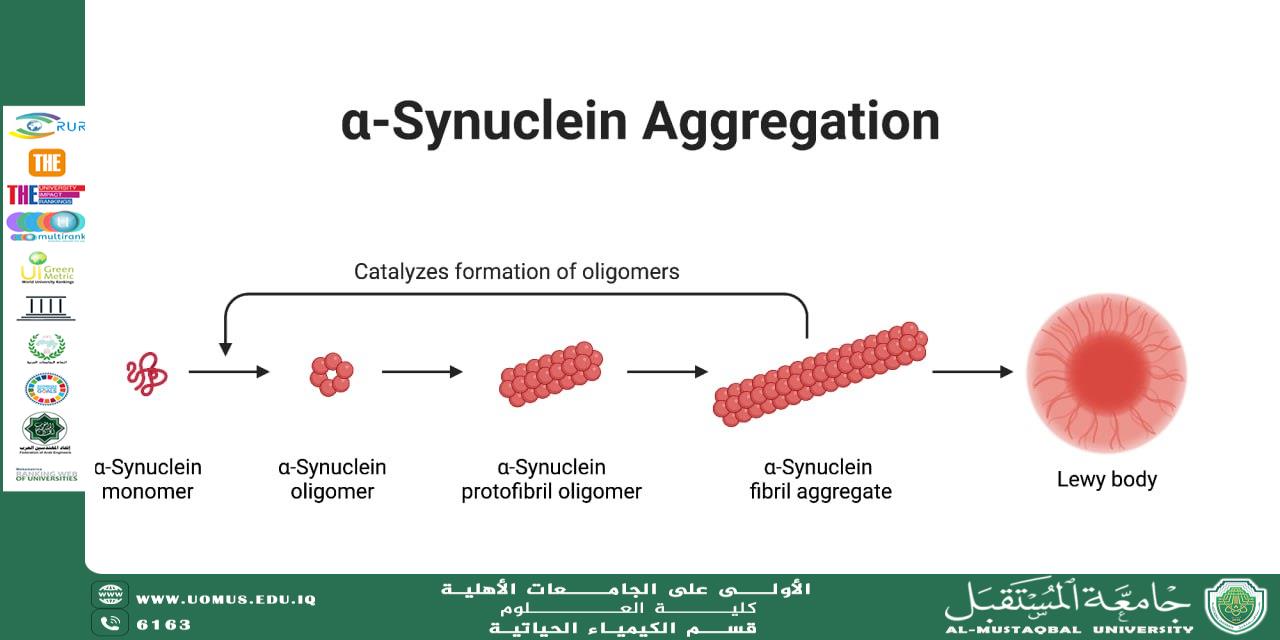
In the former, aggregation of amyloid-β peptides and tau protein is observed, whereas in the latter, pathological accumulation of α-synuclein plays a central role.
Molecular Mechanisms of Protein Aggregation
Protein aggregation generally proceeds through several sequential stages:
Conformational destabilization of the native protein structure
Formation of soluble oligomeric intermediates
Development of insoluble amyloid fibrils
Accumulation of pathological plaques or inclusions within neural tissue
Current evidence suggests that soluble oligomeric species are the most neurotoxic forms. These intermediates can:
Disrupt cellular membrane integrity
Impair mitochondrial function
Alter synaptic signaling pathways
Induce oxidative stress and inflammatory responses
Therefore, early detection and characterization of oligomeric species are critical for understanding disease progression and identifying therapeutic targets.
Technical Role of Analytical Ultracentrifugation (AUC)
Analytical Ultracentrifugation (AUC) provides a powerful, label-free approach for studying proteins in their native solution state. By measuring sedimentation behavior under high centrifugal forces, AUC enables:
Differentiation between monomers, oligomers, and higher-order aggregates
Determination of molecular weight and size distribution
Quantitative assessment of aggregation kinetics
Characterization of reversible versus irreversible aggregation processes
The Sedimentation Velocity (SV) mode is particularly useful for detecting dynamic and heterogeneous aggregation profiles, while Sedimentation Equilibrium (SE) provides accurate molecular mass determination and thermodynamic insight into association equilibria.
Unlike fluorescence-based or surface-immobilization techniques, AUC avoids structural perturbation, thereby offering highly reliable biophysical data.
Integration with Complementary Analytical Techniques
The analytical strength of AUC is significantly enhanced when combined with other structural and biophysical methods, such as:
Transmission Electron Microscopy (TEM) for direct visualization of fibrillar structures
Fluorescence spectroscopy to monitor conformational transitions
Dynamic Light Scattering (DLS) for hydrodynamic size measurement
Mass Spectrometry for molecular characterization
Such multimodal approaches provide a comprehensive understanding of aggregation pathways from structural formation to functional impact.
Early Diagnostic Implications
Recent research indicates that pathological protein aggregation may begin years—if not decades—before the onset of clinical symptoms. Consequently, sedimentation-based measurements of oligomeric species could serve as:
Early-stage molecular biomarkers
Tools for monitoring disease progression
Indicators of therapeutic response in anti-aggregation drug trials
Ongoing investigations aim to detect aggregation-prone proteins in cerebrospinal fluid (CSF) or even peripheral blood samples, potentially enabling minimally invasive early diagnosis.
Research Challenges
Despite its precision, several challenges remain:
Replicating physiological cellular environments in vitro
Maintaining physiologically relevant protein concentrations
Complex mathematical modeling of heterogeneous systems
Potential coexistence of multiple aggregate species
However, advances in computational modeling and global data-fitting algorithms have significantly improved analytical accuracy and interpretation.
Future Perspectives
Emerging research directions include:
Investigating interactions between aggregated proteins and cellular cofactors
Developing small molecules targeting early oligomeric stages
Translating AUC-based assays into clinically adaptable diagnostic tools
Applying artificial intelligence for pattern recognition in sedimentation data
These developments may shift the clinical paradigm from late-stage symptomatic diagnosis to early molecular intervention.
Analytical ultracentrifugation represents a fundamental biophysical tool for elucidating the molecular dynamics of protein aggregation in neurodegenerative diseases. By enabling precise characterization of early oligomeric species and aggregation kinetics, AUC contributes significantly to biomarker discovery and therapeutic development.
Strengthening research efforts in this field within biochemistry departments supports translational medicine initiatives and promotes early preventive strategies against neurodegenerative disorders.