Scientific Article by Ass .Prof. Mustafa Ali Al-Awadi Department of Biochemistry Titanium Dioxide (TiO₂) Nanoparticles: A Promising Technology for Pollutant Degradation Using Light
In recent years, increasing attention has been directed toward the use of nanomaterials in addressing environmental challenges. Among these materials, titanium dioxide nanoparticles (TiO₂) have gained particular importance due to their unique physical and chemical properties, which make them highly effective in photocatalytic applications. These nanoparticles have demonstrated a remarkable ability to degrade various environmental pollutants in water and air using light energy, making them a promising tool in modern environmental remediation technologies.
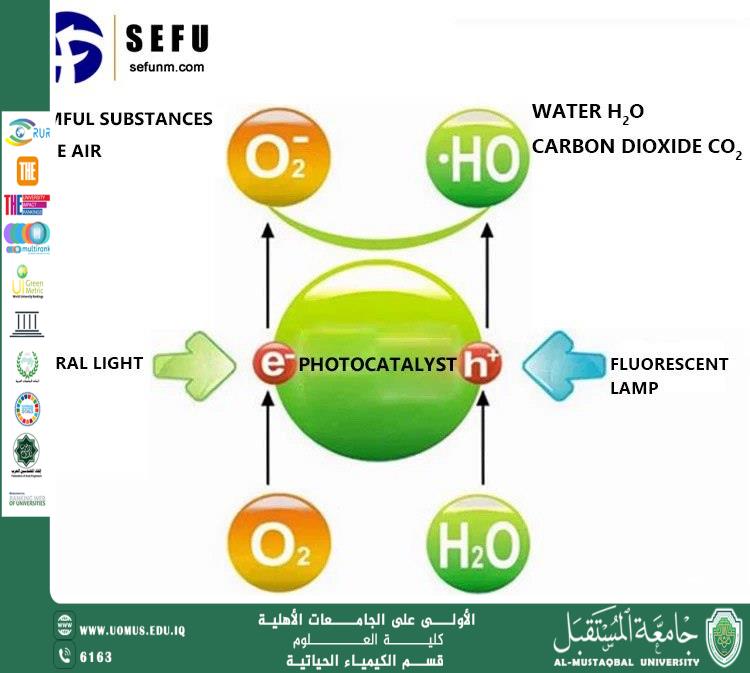
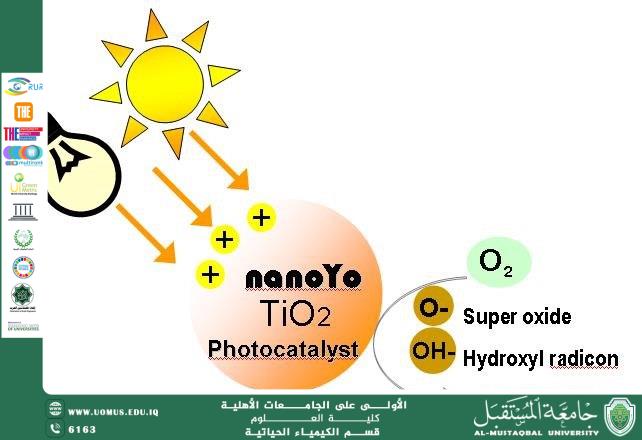
The mechanism of TiO₂ nanoparticles is based on the phenomenon of photocatalysis. When exposed to ultraviolet radiation or sunlight, electrons within the crystal structure of TiO₂ become excited and move from the valence band to the conduction band. This excitation generates electron–hole pairs that participate in chemical reactions with water and oxygen molecules present in the surrounding environment. As a result, highly reactive species such as hydroxyl radicals (•OH) are formed. These radicals are among the most powerful oxidizing agents capable of breaking down complex organic pollutants into simpler and less harmful compounds such as carbon dioxide and water.
One of the most significant advantages of TiO₂ nanoparticles is their extremely large surface area compared with conventional materials. This large surface area increases the number of active sites available for chemical reactions, thereby enhancing photocatalytic efficiency. In addition, TiO₂ nanoparticles exhibit high chemical stability, relatively low toxicity, and moderate cost, which makes them suitable for practical environmental applications. Consequently, they have been widely investigated for the treatment of industrial wastewater and the removal of organic contaminants such as synthetic dyes, pesticides, and certain pharmaceutical residues.
Furthermore, recent studies have demonstrated the potential of TiO₂ nanoparticles for air purification. These nanoparticles can be immobilized on surfaces such as glass, ceramics, or building materials, where they function as self-cleaning and pollutant-degrading coatings when exposed to light. Such coatings are capable of decomposing volatile organic compounds (VOCs) and other airborne pollutants, contributing to improved air quality in urban environments.
Current research is also focused on improving the photocatalytic efficiency of TiO₂ nanoparticles. Strategies such as doping with metal or non-metal elements and combining TiO₂ with other nanomaterials are being explored to extend their activity into the visible light region rather than relying solely on ultraviolet radiation.
Overall, TiO₂ nanoparticles represent a clear example of how nanotechnology can contribute to sustainable environmental solutions. Continued research in this field is expected to lead to more efficient and eco-friendly technologies for water and air purification, supporting global efforts to protect the environment and promote sustainable