Enzymes: Biological Catalysts in Living Systems
Enzymes are biological macromolecules, primarily proteins, that function as catalysts to accelerate chemical reactions within living cells without being consumed in the process. They are essential for maintaining life because they regulate and coordinate nearly all metabolic pathways, including catabolism, anabolism, energy production, DNA replication, and cellular signaling. Without enzymes, most biochemical reactions would occur too slowly to sustain life under physiological conditions.
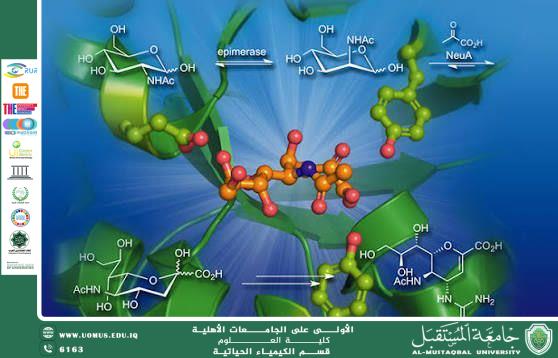
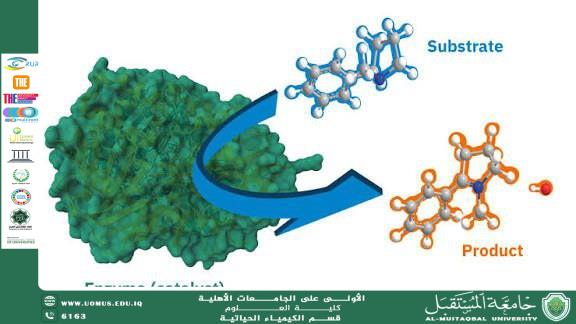
The mechanism of enzyme action depends on a specific three-dimensional region known as the active site. This site is uniquely structured to bind a particular molecule called the substrate. When the substrate binds to the active site, an enzyme–substrate complex is formed. This interaction stabilizes the transition state and lowers the activation energy required for the reaction, thereby increasing the reaction rate. Two principal models explain enzyme–substrate interaction: the lock-and-key model, which suggests a rigid complementary fit between enzyme and substrate, and the induced-fit model, which proposes that the active site undergoes a conformational change to accommodate the substrate during binding.
Enzymes exhibit remarkable specificity. Each enzyme typically catalyzes a single type of reaction or acts on a specific substrate. This specificity arises from the precise arrangement of amino acids within the active site and the overall tertiary structure of the protein. Any alteration in the enzyme’s structure, such as through extreme temperature or pH changes, can lead to denaturation, resulting in loss of enzymatic activity.
Several factors influence enzyme activity. Temperature affects molecular motion and collision frequency; most human enzymes function optimally near 37°C. pH also plays a critical role, as it influences the ionization state of amino acid residues at the active site. Additionally, substrate concentration impacts reaction velocity. According to Michaelis–Menten kinetics, the reaction rate increases with substrate concentration until it reaches a maximum velocity (Vmax), at which point all active sites are saturated. The Michaelis constant (Km) reflects the enzyme’s affinity for its substrate.
Enzyme activity can be regulated by inhibitors. Competitive inhibitors resemble the substrate and compete for binding at the active site, while noncompetitive inhibitors bind to an alternative site, causing conformational changes that reduce activity. Understanding enzyme inhibition is fundamental in pharmacology, as many drugs are designed to inhibit specific enzymes involved in disease processes.
Enzymes are classified into six major groups based on the type of reaction they catalyze: oxidoreductases, transferases, hydrolases, lyases, isomerases, and ligases. This classification highlights the diversity of biochemical reactions facilitated by enzymes in living organisms.
Beyond their biological significance, enzymes have extensive industrial and medical applications. They are used in pharmaceutical production, food processing, biotechnology, and clinical diagnostics. Measurement of specific enzymes in blood, such as liver enzymes or cardiac enzymes, serves as important diagnostic markers for various diseases.
In conclusion, enzymes are indispensable biological catalysts that sustain life by enabling efficient and regulated biochemical reactions. Their structural specificity, regulatory mechanisms, and broad applications make them central to the fields of biochemistry, molecular biology, medicine, and biotechnology.