The Sentinel Within: The Molecular Architecture of Cancer Immunosurveillance
22/02/2026 Share :
In the landscape of modern oncology, the focus has shifted from the cytotoxic destruction of cells to the sophisticated modulation of the human immune system. The "Immuno-Oncology" revolution is not merely about treating a disease; it is about restoring a biological failure of recognition.
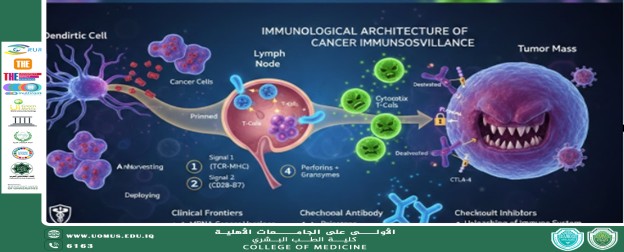
I. The Cycle of Cancer-Immunity: CycleRecognition and Priming
The battle against malignancy is a continuous cycle of surveillance. As highlighted in current molecular models, the process begins with Antigen Harvesting.
• Dendritic Cell Integration: As neoplastic cells undergo turnover or stress, they release Tumor-Associated Antigens (TAAs). Dendritic cells—the professional antigen-presenting cells (APCs)—engulf these markers.
• The "Dual Signal" Requirement: In the regional lymph nodes, these APCs present the antigens to "naive" T-cells via the Major Histocompatibility Complex (MHC). For a T-cell to transform from "naive" to "effector," it requires two signals:
1. Signal 1: The binding of the T-cell receptor (TCR) to the MHC-antigen complex.
2. Signal 2 (Co-stimulation): The interaction between B7 proteins on the APC and CD28 on the T-cell. Without this second signal, the T-cell enters a state of anergy (inactivation).
II. Deployment: The Effector Phase
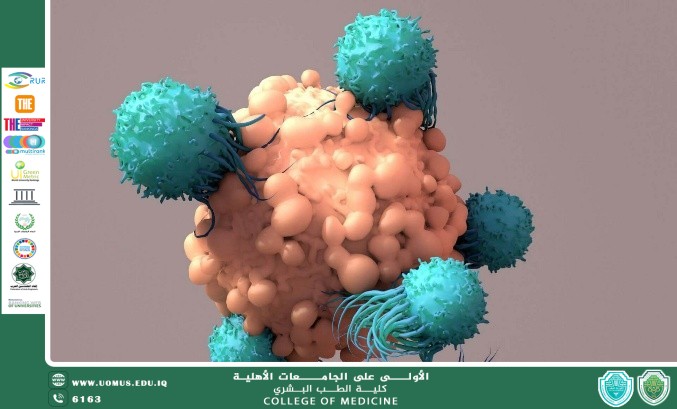
Once primed, the immune system deploys a specialized fleet to the tumor microenvironment (TME):
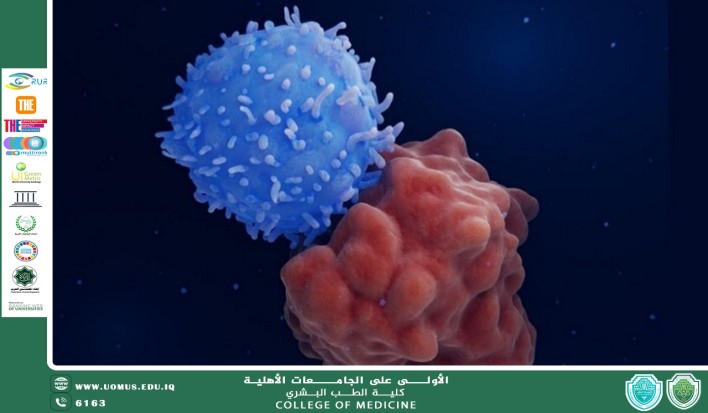
• Cytotoxic CD8+ T-cells: The primary effectors. They utilize perforins to puncture the malignant cell membrane and granzymes to initiate the caspase cascade, leading to apoptosis.
• The Role of Cytokines: Helper T-cells (CD4+) secrete Interferon-gamma ($\gamma$) and Interleukin-2, which act as biochemical amplifiers, recruiting Natural Killer (NK) cells and macrophages to the site.
III. The Mechanism of Evasion: Checkpoint Exploitation
The greatest challenge in clinical oncology is Immune Subversion. Malignant tumors are not passive targets; they actively "silence" the immune response by hijacking natural inhibitory pathways intended to prevent autoimmunity.
• The PD-1/PD-L1 Axis: Tumors often overexpress Programmed Death-Ligand 1 (PD-L1). When PD-L1 binds to the PD-1 receptor on an active T-cell, it delivers an inhibitory signal—essentially a "molecular handshake" that deactivates the T-cell.
• CTLA-4 Competition: Some tumors exploit the CTLA-4 pathway, which outcompetes the co-stimulatory signal (CD28), preventing T-cell activation at the very beginning of the cycle.
IV. Clinical Translation: The Era of Monoclonal Antibodies
Understanding these pathways has led to the development of Immune Checkpoint Inhibitors (ICIs). By using engineered monoclonal antibodies to block PD-1 or CTLA-4, we can "release the brakes" on the immune system.
Clinical Pearl for Medical Students
Unlike traditional chemotherapy, which targets rapidly dividing cells (leading to systemic toxicity), immunotherapy targets the interface between the immune system and the tumor. The side-effect profile is therefore unique, often manifesting as Immune-Related Adverse Events (irAEs) such as colitis, pneumonitis, or thyroiditis, reflecting a localized loss of self-tolerance.
Conclusion: A New Standard of Care
As we move further into 2026, the integration of Precision Immunotherapy—where a patient’s tumor is sequenced to predict which checkpoint inhibitor will be most effective—is becoming the standard. For the future physician,
the study of oncology is now inextricably linked to the study of immunology.
By: Assistant Professor Dr. Abdulhusein Mizhir Almaamuri